What is the definition of electrolyte in chemistry

David Craig
Updated on April 02, 2026
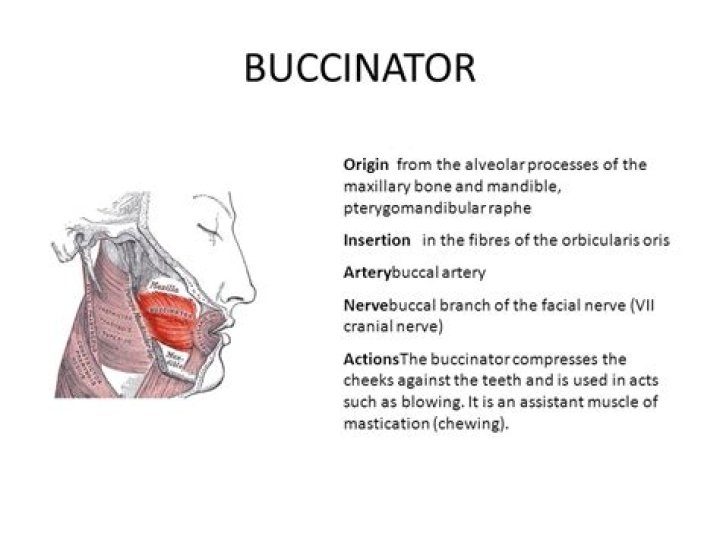
If you ask a chemistry professor to define “electrolyte,” they might say that an electrolyte is a compound which produces ions when dissolved in a solution such as water. These ions have either a positive or negative electrical charge, which is why we refer to these compounds as electro-lytes.
What is an electrolyte in chemistry simple definition?
electrolyte, in chemistry and physics, substance that conducts electric current as a result of a dissociation into positively and negatively charged particles called ions, which migrate toward and ordinarily are discharged at the negative and positive terminals (cathode and anode) of an electric circuit, respectively.
What is electrolyte in chemistry class 8?
Answer: Electrolytes are compounds that conduct electricity when they are in a solution or in a molten state. Two examples of electrolytes are NaCl (sodium chloride) and CuSO4 (copper sulphate) . Two examples of non-electrolytes are distilled water and kerosene.
What are electrolytes?
Electrolytes are minerals in your body that have an electric charge. They are in your blood, urine, tissues, and other body fluids. Electrolytes are important because they help: Balance the amount of water in your body.What is an electrolyte class 10?
An electrolyte is a substance that dissociates in water into charged particles called ions. Positively charged ions are called cations. Negatively charged ions are called anions. Simply, an electrolyte is a substance that can conduct an electric current when melted or dissolved in water.
What is strong electrolyte in chemistry?
A strong electrolyte is a solution/solute that completely, or almost completely, ionizes or dissociates in a solution. These ions are good conductors of electric current in the solution. Originally, a “strong electrolyte” was defined as a chemical that, when in aqueous solution, is a good conductor of electricity.
Why are electrolytes called electrolytes?
Substances that give ions when dissolved in water are called electrolytes. They can be divided into acids, bases, and salts, because they all give ions when dissolved in water. These solutions conduct electricity due to the mobility of the positive and negative ions, which are called cations and anions respectively.
What are electrolytes and Nonelectrolytes 8?
An electrolyte contains mobile ions which move from one electrode to another electrode. Thus, it conducts current. All ionic compounds are electrolytes. A nonelectrolyte is a compound that does not conduct an electric current in either aqueous solution or in the molten state.What is the electrolyte used?
Electrolytes are chemicals that conduct electricity when dissolved in water. They regulate nerve and muscle function, hydrate the body, balance blood acidity and pressure, and help rebuild damaged tissue. The muscles and neurons are sometimes referred to as the “electric tissues” of the body.
What is electrolyte and its types?Electrolyte: A substance that dissociates into ions in solution and acquires the capacity to conduct electricity. Sodium, potassium, chloride, calcium, and phosphate are examples of electrolytes, informally known as lytes.
Article first time published onWhat are electrolytes and their types?
Strong Electrolytesstrong acidsHCl, HBr, HI, HNO3, HClO3, HClO4, and H2SO4strong basesNaOH, KOH, LiOH, Ba(OH)2, and Ca(OH)2saltsNaCl, KBr, MgCl2, and many, many moreWeak Electrolytesweak acidsHF, HC2H3O2 (acetic acid), H2CO3 (carbonic acid), H3PO4 (phosphoric acid), and many more
What is electrolyte class 11?
Hint: Electrolyte is a liquid or gel which contains ions and can be decomposed by electrolysis (Electrolysis is the Chemical decomposition of substances by passage of electric current). It produces an electrically conducting solution when dissolved in polar solvent such as water.
What is electrolyte in chemistry 12th class?
Electrolyte :It is a compound which either in aqueous solution or in the molten state allows an electric current to pass through it and is accompanied by discharge of ions and finally into neutral atoms at the two electodes. For example : Hydrochloric acid.
What is the electrolyte class 12?
CBSE NCERT Notes Class 12 Chemistry Electrochemistry. A solute or substances that completely ionize or dissociates in a solution are known as strong electrolyte. These ions are good conductors of electricity in the solution. For example, HCl, HBr, HI, HNO3, NaOH, KOH, etc.
What are the 3 main electrolytes?
The major electrolytes: sodium, potassium, and chloride.
What is the difference between electrolytes and minerals?
Electrolytes are typically minerals, whereas minerals may or may not be electrolytes. Further, electrolytes can be either “essential minerals”, or they can be non-essential (not needed by the body), and some electrolytes are even toxic!
What is strong electrolyte and example?
Substances that completely dissociates into ions are called strong electrolytes. Example: Sodium chloride, potassium chloride, lead bromide, sodium hydroxide, potassium hydroxide, hydrochloric acid, nitric acid, sulfuric acid, etc.
What is the difference between strong and weak electrolytes?
A strong electrolyte is a solution in which a large fraction of the dissolved solute exists as ions. … A weak electrolyte is a solution in which only a small fraction of the dissolved solute exists as ions.
Which is the weak electrolyte?
Weak Electrolyte Examples HC2H3O2 (acetic acid), H2CO3 (carbonic acid), NH3 (ammonia), and H3PO4 (phosphoric acid) are all examples of weak electrolytes. Weak acids and weak bases are weak electrolytes. In contrast, strong acids, strong bases, and salts are strong electrolytes.
What is electrolyte in electroplating?
Generally, one of the electrodes is made from the metal we’re trying to plate and the electrolyte is a solution of a salt of the same metal. So, for example, if we’re copper plating some brass, we need a copper electrode, a brass electrode, and a solution of a copper-based compound such as copper sulfate solution.
What are electrolytes in water?
What Is Electrolyte Water? Electrolytes are minerals that conduct electricity when dissolved in water. They’re distributed through the fluid in your body and use their electrical energy to facilitate important bodily functions (1).
What is called non electrolyte?
Nonelectrolytes are compounds that do not ionize at all in solution. … Glucose (sugar) readily dissolves in water, but because it does not dissociate into ions in solution, it is considered a nonelectrolyte; solutions containing glucose do not, therefore, conduct electricity.
What are Nonelectrolytes?
Definition of nonelectrolyte : a substance that does not readily ionize when dissolved or melted and is a poor conductor of electricity.
Is electrolyte a metal?
An electrolyte is : a cell. a metal. a liquid that conducts electricity.
What are electrolytes made of?
Electrolytes are salts and minerals, like sodium, potassium, calcium, magnesium and chloride, in the body that maintain fluid balance and blood pressure.
Which of these is an electrolyte?
A substance that, when added to water, renders it conductive, is known as an electrolyte . A common example of an electrolyte is ordinary salt, sodium chloride. Solid NaCl and pure water are both non-conductive, but a solution of salt in water is readily conductive.