What is Siliq used for

Andrew Campbell
Updated on April 13, 2026
Siliq is a brand-name prescription medication. It’s used to treat moderate to severe plaque psoriasis in adults. Plaque psoriasis is one of many kinds of psoriasis. Siliq is a systemic treatment, which means it works throughout your body.
How long does Siliq take to work?
It may take up to 16 weeks before your symptoms improve. Keep using the medication as directed and tell your doctor if your symptoms do not improve after 12 weeks of using Siliq.
Is Siliq approved for psoriatic arthritis?
The U.S. Food and Drug Administration has approved brodalumab (Siliq) for the treatment of adults and moderate-to-severe plaque psoriasis. Brodalumab, which is administered as an injection, is a monoclonal antibody that neutralizes IL-17 receptor type A – an important cytokine in psoriatic arthritis.
What kind of drug is Siliq?
This medication is used to treat plaque psoriasis. Brodalumab belongs to a class of drugs known as monoclonal antibodies. It works by blocking a certain natural protein in your body (interleukin-17A) that may cause inflammation and swelling.How do you administer Siliq?
- Set up. First, set up all your supplies.
- Select. Select an injection site, and with clean hands, wipe the site with an alcohol swab. …
- Squeeze. Squeeze the injection site to create a firm surface.
- Inject SILIQ.
Who manufactures Siliq?
Valeant Pharmaceuticals International, Inc.
Is Siliq safe?
This drug has a boxed warning. This is the most serious warning from the Food and Drug Administration (FDA). A boxed warning alerts doctors and patients about drug effects that may be dangerous. Some people who’ve taken Siliq have had suicidal thoughts and behavior, and even died by suicide.
What is Duobrii for?
This medication is a combination product used to treat a certain skin condition (psoriasis). It contains 2 ingredients: halobetasol and tazarotene. Halobetasol is a very strong (super-high potency) corticosteroid. It helps reduce the itching, redness, scaling, and swelling that can occur with this condition.How often is Tremfya taken?
TREMFYA is administered by subcutaneous injection. The recommended dose is 100 mg at Week 0, Week 4, and every 8 weeks thereafter. TREMFYA is administered by subcutaneous injection. The recommended dose is 100 mg at Week 0, Week 4, and every 8 weeks thereafter.
Is Tremfya used for psoriatic arthritis?TREMFYA® is a prescription medicine used to treat adults with active psoriatic arthritis.
Article first time published onWhat is the brand name for ustekinumab?
Monoclonal antibodyTrade namesStelaraOther namesCNTO 1275AHFS/Drugs.comMonographMedlinePlusa611013
Who owns Cosentyx?
Secukinumab, sold under the brand name Cosentyx, is a human IgG1κ monoclonal antibody that binds to the protein interleukin (IL)-17A, and is marketed by Novartis for the treatment of psoriasis, ankylosing spondylitis, and psoriatic arthritis.
What is Brodalumab used for?
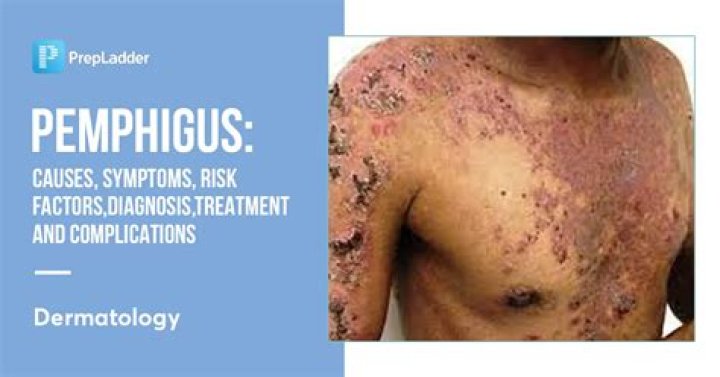
Brodalumab injection is used to treat moderate to severe plaque psoriasis (skin disease in which red, scaly patches form on some areas of the body) in people whose psoriasis is too severe to be treated by topical medications alone and who have not been treated successfully with other medications.
Who makes Tildrakizumab?
ILUMYA (tildrakizumab-asmn) is a subcutaneous infusion indicated for the treatment of adults with moderate-to-severe plaque psoriasis. The drug was developed by Schering-Plough, which merged with US-based drug maker Merck & Co in 2009. Sun Pharma acquired worldwide rights for ILUMYA from Merck & Co for $80m.
Is Taltz a monoclonal antibody?
Ixekizumab, sold under the brand name Taltz, is an injectable medication for the treatment of autoimmune diseases. Chemically, it is a form of a humanized monoclonal antibody.
Who makes Bimekizumab?
This drug is being developed by Belgian pharmaceutical UCB.
What is the mechanism of action of apremilast?
The full mechanism of action of this drug is not fully established, however, it is known that apremilast is an inhibitor of phosphodiesterase 4 (PDE4), which mediates the activity of cyclic adenosine monophosphate (cAMP), a second messenger.
Is Skyrizi humanized?
Risankizumab, sold under the brand name Skyrizi, is a humanized monoclonal antibody targeting interleukin 23A (IL-23A). Risankizumab is part of a collaboration between Boehringer Ingelheim and AbbVie.
What is Tremfya prescribed for?
This medication is used to treat plaque psoriasis and psoriatic arthritis. Guselkumab belongs to a class of drugs known as monoclonal antibodies. It works by blocking a certain natural substance in your body (interleukin-23) that may cause inflammation and swelling.
Is Tremfya better than Stelara?
Patients receiving TREMFYA had a significantly greater number of visits from week 28 to week 40 with an Investigator’s Global Assessment (IGA) score of 0 or 1 and ≥2-grade improvement from week 16 vs patients receiving STELARA (P<0.001).
Does Tremfya cause liver damage?
Elevated liver enzymes were reported more frequently in the TREMFYA group (2.6%) than in the placebo group (1.9%). Of the 21 subjects who were reported to have elevated liver enzymes in the TREMFYA group, all events except one were mild to moderate in severity and none of the events led to discontinuation of TREMFYA.
Is Duobrii a steroid?
Duobrii Lotion contains a combination of halobetasol and tazarotene. Halobetasol is a highly potent steroid. Tazarotene is a compound similar to vitamin A. Duobrii Lotion is a prescription medicine used to treat plaque psoriasis (raised, silvery flaking and redness of the skin) in adults.
How often can you use Duobrii?
DUOBRII™ (halobetasol propionate and tazarotene) Lotion, 0.01%/0.045% is indicated for the topical treatment of plaque psoriasis in adults. Apply a thin layer of DUOBRII Lotion once daily to cover only affected areas and rub in gently.
How long does it take for Duobrii to work?
12 weeks of results with 8 weeks of treatment Almost clear = some plaques with fine scales, faint pink/light redness on most plaques, slight or barely noticeable elevation of plaques above normal skin level.
Which is better Taltz or Tremfya?
Taltz was superior to Tremfya in providing rapid and visible skin clearance (PASI 75) at week 2. In an NRI analysis of IXORA-R (Taltz n=520; Tremfya n=507), 23% * of patients receiving Taltz achieved PASI 75 at week 2 vs 5% of patients receiving Tremfya.
Is there a link between psoriasis and high blood pressure?
In fact, more than half (54 percent) of the psoriasis patients had high blood pressure. Results from a patient database in the United Kingdom revealed that people with moderate or severe psoriasis were 20 and 48 percent, respectively, more likely to have uncontrolled high blood pressure.
Can Tremfya cause lymphoma?
Most clinical trials investigating Tremfya excluded people from participating in the trial if they had current cancer or a previous history of cancer. There is also evidence to show that people with psoriasis have an inherent increased risk of developing several different malignancies including NMSC and lymphoma.
Is ustekinumab a steroid?
Is STELARA® a steroid? No. STELARA® is not a steroid. STELARA® targets an underlying cause of plaque psoriasis and psoriatic arthritis—an overactive immune system.
How long does ustekinumab take to work?
How long will it take to work? Everyone responds differently when taking a new medicine, and ustekinumab doesn’t work for everyone. You may feel better as early as three weeks after taking ustekinumab, but most people who respond to ustekinumab start feeling better within six weeks. In some people it could take longer.
What is Crohn's disease and what does it do to you?
Crohn’s disease is a type of inflammatory bowel disease (IBD). It causes inflammation of your digestive tract, which can lead to abdominal pain, severe diarrhea, fatigue, weight loss and malnutrition.
What is Cosentyx used to treat?
COSENTYX is a biologic that helps reduce the symptoms of ankylosing spondylitis (AS), like back pain and morning stiffness. It’s the first biologic of its kind to treat AS by targeting a molecule called IL-17A.